Author: Erica Woodahl, PhD on February 15, 2021 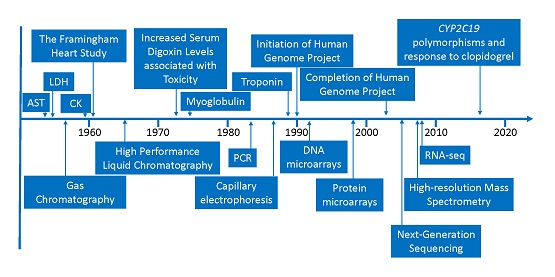
Clinical and Translational Science (CTS) is pleased to present a recent position paper by Oni‐Orisan et al. on behalf of the Biomarkers & Translational Tools Community of the American Society for Clinical Pharmacology & Therapeutics (ASCPT): “Leveraging innovative technology to generate drug response phenotypes for the advancement of biomarker‐driven precision dosing.” Precision dosing is a component of precision medicine that aims to optimize the right dose for patients to increase therapeutic efficacy and minimize adverse events. The Biomarkers & Translational Tools Community Working Group brought together a diverse group of authors from academia and industry to offer a multidisciplinary position paper on identifying biomarkers of drug response to tailor dosing.
Looking beyond traditional approaches such as randomized control trials and observational studies—which have been useful in identifying pharmacogenetic biomarkers—the authors discuss innovative approaches to gathering drug response biomarkers from real-world data sources. These approaches (electronic health record-linked biobanks, direct-to-consumer genetic testing, social media, mobile devices, and machine learning) have been used outside of biomedical research to great success, and the authors discuss opportunities for these approaches to drive knowledge in precision dosing. Partnerships between academia and industry will be key to advancing biomarker-driven precision dosing.

The comment feature is locked by administrator.