Author: Ming Huang, PhD on January 12, 2023 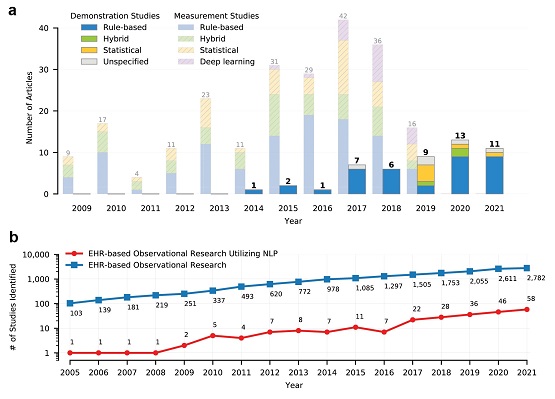
An increasing number of studies have reported using natural language processing (NLP) to assist observational research by extracting clinical information from electronic health records (EHRs). Currently, no standardized reporting guidelines for NLP-assisted observational studies exist. The absence of detailed reporting guidelines may create ambiguity in the use of NLP-derived content, knowledge gaps in the current research reporting practices, and reproducibility challenges. Clinical and Translational Science (CTS) recently published our article entitled “Recommended Practices and Ethical Considerations for Natural Language Processing-Assisted Observational Research: A Scoping Review,” which examined the current reporting practice of NLP-empowered observational studies based on the RITE (Reproducible, Implementable, Transparent, and Explainable)-FAIR (Findable, Accessible, Interoperable, and Reusable) principles.
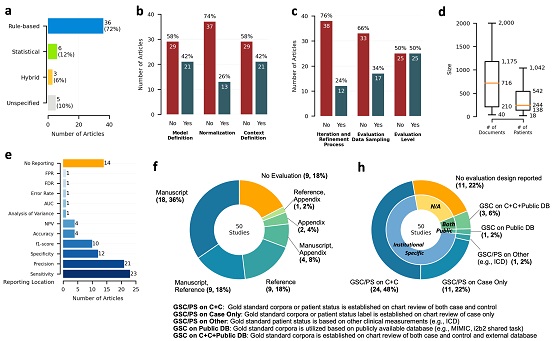
The study revealed a high variation regarding the reporting practices, such as inconsistent use of references for measurement studies, variable reporting location, and different granularity of NLP methodology and evaluation details. To align with the six guiding principles released by WHO to guide the ethical use of AI for health, we outlined two key perspectives to promote the wide adoption and utilization of NLP solutions in clinical research: 1) ensuring process transparency and scientific rigor, and 2) promoting open NLP communities through team science collaboration.

The comment feature is locked by administrator.