Author: Nina Isoherranen, PhD on August 10, 2017 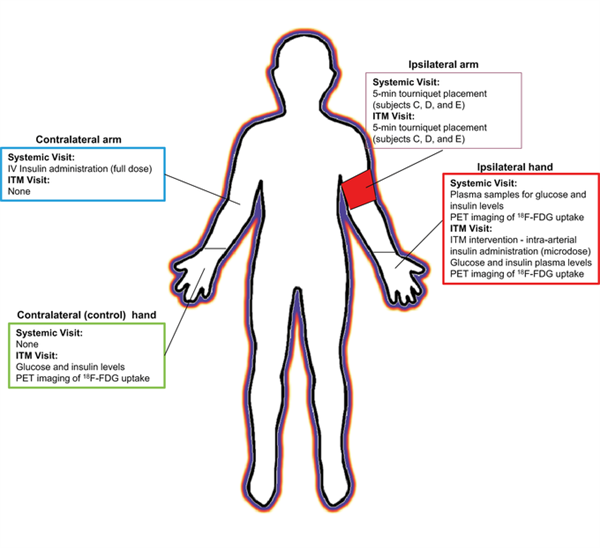
Microdosing, an approach which uses doses 1/100th of the anticipated minimum effective dose or a dose of 100 mg, whichever is lower, is a well-established method to study pharmacokinetic characteristics and drug-drug interaction susceptibility of drug candidates and drugs for which significant increases in exposure may pose a safety concern. In the article published in Clinical and Translational Science in July 2017, Burt et al. provide a proof of concept study of the combination of microdosing concepts with intra-target dosing. This exciting, first of its kind, study involved insulin microdosing into the radial artery (intra-target) and dosing of a full therapeutic dose of insulin intravenously to healthy volunteers on two separate occasions. Insulin pharmacokinetics was measured to evaluate systemic exposures. Plasma glucose in the ipsilateral and contralateral arms to the microdosing and 18F- fluorodeoxyglucose uptake were evaluated as pharmacodynamic measures to delineate local and systemic effects. This study is highly innovative and an important proof of concept study. It demonstrates that intra-target microdosing can provide similar pharmacodynamic effects as systemic full dose exposure while keeping systemic exposures at the level of a microdosing paradigm. Such approaches have great potential to increase the impact of “phase 0” studies in drug development and in future evaluation of drug efficacy and pharmacokinetics in vulnerable populations. Via establishing the safety and feasibility of intra-target microdosing, this study has laid the groundwork for larger studies in the future that allow simultaneous evaluation of efficacy at the target site and pharmacokinetic characteristics.
Image by Burt, et al. Clin. Trans. Sci., doi: 10.1111/cts.12477, is licensed under CC BY-NC-ND 4.0. ©2017 The authors.

The comment feature is locked by administrator.