Author: William Jusko, PhD, and Artur Świerczek, PhD on November 20, 2023 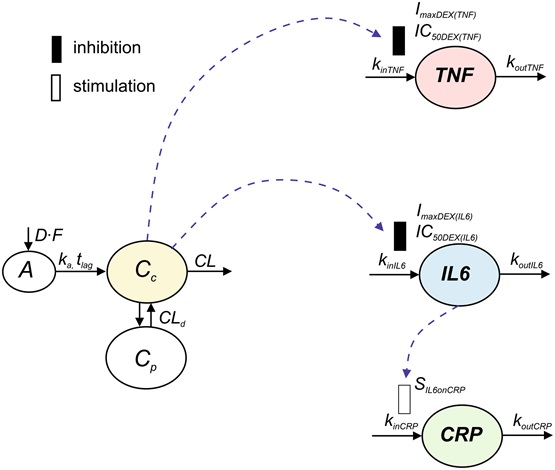
The employment of mechanistic pharmacokinetic/pharmacodynamic (PK/PD) modeling and simulation in translational science offers a critical pathway for bridging the gap between experimental research and clinical application. Our recent study, “Anti-Inflammatory Effects of Dexamethasone in COVID-19 Patients: Translational Population PK/PD Modeling and Simulation,” leverages this approach to explore the efficacy of dexamethasone, a key medication in the anti-inflammatory arsenal against COVID-19. By using both pre-clinical and clinical data, we developed a population PK/PD model of dexamethasone effects in COVID-19-related cytokine storm. Using this model, we performed simulations of the expected efficacy of four dosing regimens of this drug in reducing critical inflammatory biomarkers, i.e., TNFα, IL-6, and CRP.
Dexamethasone has become an important element of COVID-19 treatment in patients requiring oxygen therapy. Our research reveals that daily doses of 6 or 12 mg dexamethasone for 10 days are required for substantial reductions in TNFα, IL-6, and CRP concentrations. This study not only reaffirms the current recommended dosing regimen of dexamethasone in the treatment of COVID-19 patients, which is 6 mg daily for 10 days, but also uncovers the potential advantages of the twice higher dose.
This study highlights the increasing role of mechanistic PK/PD modeling and simulation in advancing translational science. Our study underscores the considerable potential of this approach in accelerating the journey from laboratory bench to bedside, guiding more precise drug dosing, and improving patient outcomes, especially in the context of the ongoing global health crisis.

The comment feature is locked by administrator.