Author: Robert Grossman, PhD, and Jake Vinson, MHA on August 28, 2025 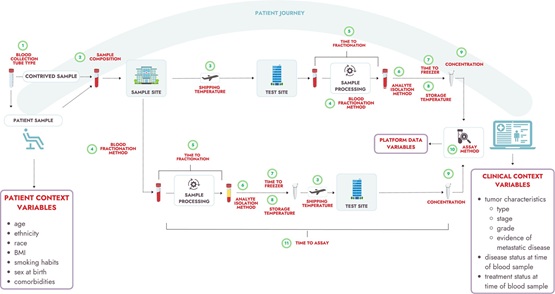
Liquid biopsies are tests for detecting a cancer-associated signal in a body fluid sample to improve outcomes for patients with cancer. BLOODPAC is a not for profit consortium that works to accelerate the development, validation, and accessibility of liquid biopsies. More than 40 BLOODPAC members have contributed data to the BLOODPAC Data Commons, a cloud-based data platform that supports liquid biopsy research and standardization efforts. An analysis of the contributed data led members to standardize the fields of data submitted to data platforms such as the BLOODPAC Data Commons. This is a critical step toward ensuring consistency and supporting comparability across liquid biopsy studies. These data harmonization efforts aim to facilitate the regulatory review process for liquid biopsy assays, making them more accessible to cancer patients and ultimately improving patient care.
The initial effort of what is now the Recommended Data Elements Working Group led to a 2020 publication recommending 11 pre-analytical attributes that should be collected for liquid biopsy assays, with an emphasis on contributors to the BLOODPAC Data Commons or similar repositories. In this article, we extend that work by recommending 22 clinical context data elements and 10 patient context data elements, along with several administrative variables. This standardization is important for enabling cross-assay comparisons and joint analyses through the BLOODPAC Data Commons or other public databases. All variables are either required or recommended for data submitted to the BLOODPAC Data Commons or similar public databases.
A key challenge is relating a liquid biopsy test result to the clinical context in order to provide a robust indicator for disease monitoring and therapeutic efficacy. The 22 clinical variables proposed represent a concise set of criteria to evaluate the type, timing, and extent of a patient’s disease all with the end goal of helping to determine where the patient is in their treatment journey. For example, clinical context data elements proposed include tumor type, stage, grade, date of initial diagnosis, and whether clinicopathological sampling and imaging show evidence of metastatic disease.
The group also identified a set of patient demographic traits to be submitted alongside clinical data. These elements are readily available, well-defined, and provide meaningful insights into disease detection and health outcomes. They include age, ethnicity, race, BMI, tobacco smoking history and amount, sex at birth, self-identified gender, fasting status, and clinically validated comorbidities known to influence liquid biopsy detection and analyte concentrations.
The establishment of these data elements represents a key step toward data harmonization and standardization. BLOODPAC plans to revisit these elements to ensure alignment with the rapidly evolving field. BLOODPAC welcomes comments and suggestions to improve development and utility of liquid biopsies. Please contact us at www.bloodpac.org with inquiries or suggestions!

The comment feature is locked by administrator.