Author: Michael Pacanowski, PharmD, MPH on April 19, 2023 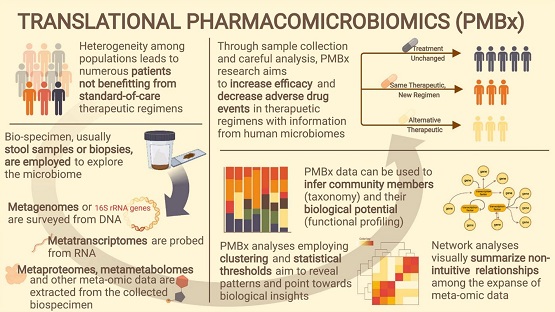
Clinical pharmacology is often viewed as what a drug does to the body and what the body does to a drug. On and in our bodies is a universe of microbes, and each microorganism has its own cellular machinery that can interact with drugs, too. However, the trillions of cells that make up an individual’s unique flora of bacteria, fungi, and other microscopic organisms are often an overlooked source of drug exposure or response variability. Moreover, medicines can tilt the delicate balance of the microbiome, which can adversely affect health or, for some conditions, be the target treatment.
Recently Clinical and Translational Science (CTS) published two tutorials that provide in-depth reviews on the state of knowledge for “pharmacomicrobiomics” and how to conduct microbiome studies. In the December 2022 issue, Dodd and Caan offer a detailed orientation to the human microbiome, factors that lead to interindividual variability in microbial communities, and the ways that microbes can influence drug metabolism. The authors then provide an overview of the various models in which microbiome effects on drug response can be investigated, focusing mainly on nonclinical approaches. Following along in the translational continuum, in the October 2022 issue, Steiner et al. describe how to study the human microbiome in the clinic. Several issues warrant consideration, including the design, inclusion criteria, clinical data collection (particularly diet and medication use), specimen sampling, assay methods, and data analysis. (This paper covers many important considerations for authors and peer-reviewers to consider!) For those who are interested in adding microbiome assessments to their studies, or perhaps just want to learn a bit more about the science, both papers are excellent resources.
Some interesting, additional microbiome studies published in CTS last year:

The comment feature is locked by administrator.