Author: Lindsay M. Henderson, Pharm.D., Ph.D. on May 30, 2023 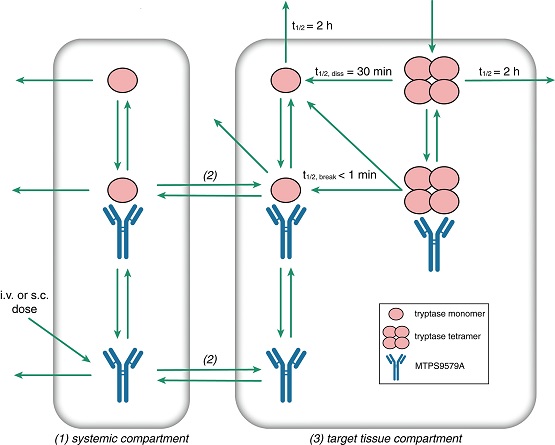
Despite multiple available therapies, many patients with asthma still have symptoms that are not well controlled. Most current therapies for asthma target mediators of type 2 inflammation (e.g., interleukins), but these therapies do not provide adequate relief for many patients with type 2-independent asthma. MTPS9579A is a monoclonal antibody that targets the mast cell, a central player in type 2-independent asthma. Specifically, MTPS9579A inhibits tryptase, a major mast cell product that induces airway smooth muscle contraction and hyperreactivity, by dissociating active tetramers into inactive monomers.
In a study recently published in Clinical and Translational Science (CTS), Lindsay Henderson and colleagues describe the development of a mechanistic pharmacokinetic/pharmacodynamic (PK/PD) model that incorporates the local tissue distribution and local target binding kinetics of MTPS9579A to represent the unique mechanism of irreversible dissociation of active tryptase tetramers. This PK/PD model predicts almost complete suppression (i.e., >95%) of active tryptase in the airway of patients with moderate-to-severe asthma with MTPS9579A doses of 900 mg and greater, administered intravenously once every 4 weeks. This dose prediction sets the stage for clinical testing of the tryptase inhibition hypothesis in patients with asthma.
This article should be of great interest to clinical pharmacologists and pharmacometricians who employ model-informed drug development approaches in their decision making and development strategies. PK/PD modeling enables the integration of disease pathophysiology with nonclinical and clinical data to characterize a molecule’s exposure-response relationship. In this study, we highlight how PK/PD modeling can be utilized to quantitatively assess the relationship between PK and biomarkers in the target tissue to help inform dose selection for future proof-of-concept clinical studies.

The comment feature is locked by administrator.