Author: Stephan Chalon, MD, PhD, and Naomi Richardson on February 27, 2026 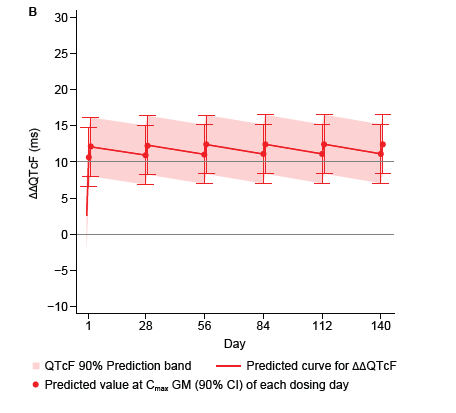
Figure: Drug-specific predicted QTcF effect based on the concentration–ΔQTcF model for PQP. Predicted ΔΔQTcF at the simulated Cmax following coadministration of PYR+PQP once daily for 2 days on the first day of each month for 6 months dosed by bodyweight: PYR 540 mg (50–<65 kg) or 720 mg (≥65 kg); PQP 960 mg (50–<75 kg) or 1,280 mg (≥75 kg). Data are presented as predicted values at geometric mean Cmax (points with 90% CI) and predicted curves with 90% prediction intervals.
A randomized, double-blind, placebo-controlled Phase I study in 37 healthy adults of Black African origin assessed the cardiac safety of combining pyronaridine (PYR) and piperaquine (PQP), two oral long-acting antimalarial agents with potential for monthly chemoprevention. PQP is known to produce an exposure-dependent prolongation of the Fridericia corrected QT interval (QTcF), while PYR has not demonstrated clinically significant QT effects at therapeutic doses. Using matched high-resolution electrocardiogram and pharmacokinetic data, investigators developed a concentration–QTc model to quantify the impact of each drug alone and in combination.
PQP alone increased the placebo- and baseline-adjusted QTcF beyond the 10-msec regulatory threshold, whereas PYR alone did not. Co-administration produced a modest additional increase, attributable mainly to a rise in PQP peak concentrations, but the effect plateaued at higher exposures. Importantly, model-based simulations of a monthly two-day dosing regimen predicted no cumulative QTcF prolongation over six months (see Figure). These findings indicate that, when administered in the fasted state, the combination’s cardiac risk profile was comparable to PQP alone.
From a translational perspective, these results show how well-designed Phase I studies can efficiently provide the necessary data for concentration–QTc modelling acceptable to regulatory agencies, even in the case of combination therapies. In this case, the model also allowed investigation of the impact on QTcF of alternative dosing regimens relevant to clinical use. Concentration–QTc modeling thereby provides early, regulatory-aligned evidence on cardiac safety, enabling informed decision-making before larger clinical studies.

The comment feature is locked by administrator.